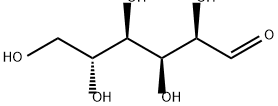
CAS No.: 50-99-7 Formula: C6H12O6
Weight: 180.156
Synonyms: D(+)-Glucose; β-D-Glucose anhydrous; Dextrose anhydrous; Dextrose; Glucose anhydrous; Glucose (industrial grade);
In medicine, it can be formulated into oral liquid or intravenous injection as nutritional supplement, used as sweetener in food industry, used in biological culture medium preparation and pharmaceutical industry, and also used as reducing agent, as reducing agent, sugar, and also used in biological culture. Glucose is a six-carbon sugar containing an aldehyde group with a molecular formula of C6H12O6. It is the most important monosaccharide in living organisms. Its main function is to provide the energy required for the body's physiological activities. It is known that 1mol of glucose is completely oxidized to release energy of 2870kJ (686kCal), accounting for 70% to 80% of the total energy required by the body. In addition to supplying energy, glucose is also a carbon source for human biosynthesis and is an important precursor for lipid synthesis; it also provides a framework for the synthesis of essential amino acids in the body. Under normal circumstances, the concentration of glucose (blood sugar) in the blood is in a dynamic balance, and the body has a set of mechanisms for regulating blood sugar concentration by nerves, liver, hormones, and kidneys. The normal fasting blood glucose concentration is 3.8 to 6.1 mmol/L.