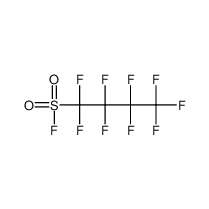
Nonafluorobutanesulfonyl fluoride
CAS No.: 375-72-4 Formula: C4F10O2S
Weight: 302.09100
Synonyms: 1,1,2,2,3,3,4,4,4-Nonafluorobutane-1-sulfonyl fluoride; Nonafluorobutanesulfonyl Fluoride; Nonafluoro-1-butanesulfonyl Fluoride; Perfluoro-1-butanesulfonyl Fluoride;
Perfluorobutanesulfonyl fluoride (nonafluorobutanesulfonyl fluoride, NfF) is a colorless, volatile liquid that is immiscible with water but soluble in common organic solvents. It is prepared by the electrochemical fluorination of sulfolane. NfF serves as an entry point to nonafluorobutanesulfonates (nonaflates), which are valuable as electrophiles in palladium catalyzed cross coupling reactions. As a perfluoroalkylsulfonylating agent, NfF offers the advantages of lesser cost and greater stability over the more frequently used triflic anhydride. The fluoride leaving group is readily substituted by nucleophiles such as amines, phenoxides, and enolates, giving sulfonamides, aryl nonaflates, and alkenyl nonaflates respectively. However, it is not attacked by water (in which it is stable at pH<12). Hydrolysis by barium hydroxide gives Ba(ONf)2, which upon treatment with sulfuric acid gives perfluorobutanesulfonic acid and insoluble barium sulfate.